Organ Manufacturing - Introduction
Organ manufacturing is the process of creating a bioartificial organ outside of the human body. For the longest time it was somewhat of a sci-fi goal, but with recent advancements in stem cells, genetic engineering and manufacturing technologies, it has become a reality that is not too far away.
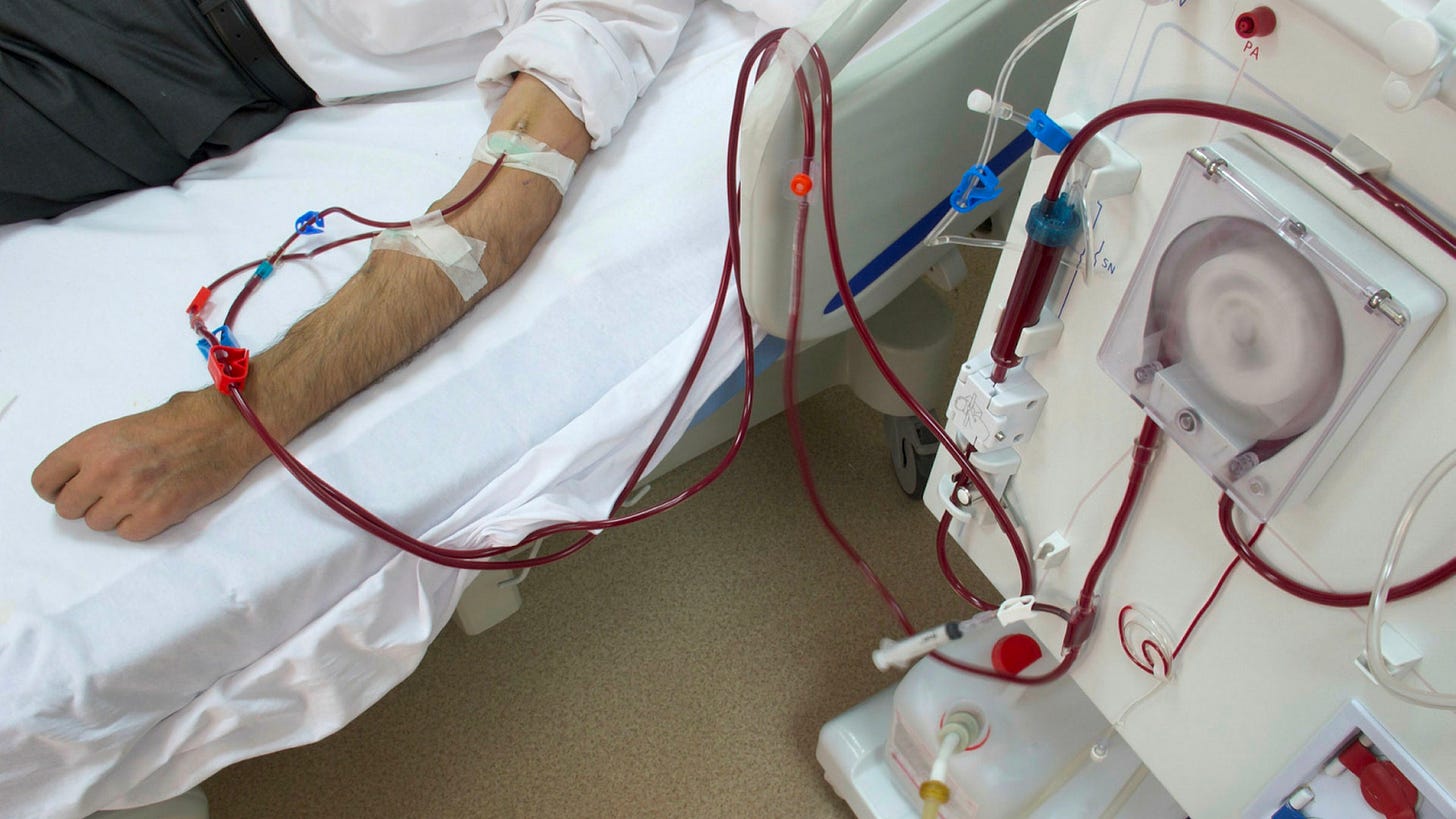
You might think artificial organs are a thing of the future, but they’re actually pretty common today! Artificial organs of today are usually mechanical, however, and therefore don’t really replace the full functionality of your own organs. Some well known examples are dialysis machines and mechanical ventilators.
Man on dialysis machine - Source
This newsletter’s focus, if you haven't already guessed, will be on bioartificial organs that don’t require an external power source.
Why is this space interesting?
Every year, more than 200,000 people globally are officially on the waiting list for organ transplantation - this number doesn’t include the number of people needing a transplant - with many more millions of people waiting on a tissue transplant. People fortunate to be on transplant lists can wait anywhere between 3-10 years, or in some cases, die waiting. These transplants save lives and are the best chance of giving people a high quality of life.
Beyond the fact that it saves lives, it can also prevent life ever getting to that state by allowing for really accurate disease modeling, studying, and even drug testing.
On a more weirder note, I also think manufactured organs could drastically reduce biotech’s barrier to entry. Imagine if people motivated or curious enough to venture into biotech without formal training were able to learn and experiment on real organs, without putting lives at risk? It would open up a whole new world of biotech founders, products and discoveries. It would be the equivalent of making computers accessible to the masses. The ripple effect of this could be insane!
All this is to say that there are really worthwhile opportunities that could come from making this a reality, so we should be working hard to do so.
What bioartificial organs exist today?
Quite a few, but not enough! Around 7 artificial bladders have been created and successfully implanted in patients. These artificial bladders were created from the patient's own cells, meaning that the patient will not have to take immunosuppressants for the rest of their lives, as their body does not see it as a foreign object. Although this is an impressive feat, we are still a long way away from doing so with kidneys (the most in demand organ for transplantation), hearts, brains and other complex organs (these organs are complex because they contain a wide variety of cells that have to be in particular places to function as intended).
Putting aside the complexity of these organs, is the additional difficulty of creating these cells. In the ideal scenario you would want to use the patient’s own cells, to avoid rejection. To do so, stem cell technology has to be used, and although we have made major advancements in this field, particularly with induced pluripotent stem cells, they still do not always perform at the same efficacy as the original cell - which can be very dangerous if put into a patient.
Let’s take it a step further and assume that we perfectly solve the problem of creating the cells and putting the cells in the right place. None of this, unfortunately, will matter if these cells aren’t alive, or can’t be kept alive. And that brings us to the big thorny problem in organ manufacturing, vascularisation. Our vascular system is our network of different blood vessels that provide nutrients (such as oxygen and salts) to our cells to keep them alive. I could explain why vascularisation is so hard, but this reddit thread does a much better (high level) job of explaining why vascularisation is so difficult. Vascularisation on its own is a *whole* topic, so you can expect to see its own dedicated post sometime soon!
That being said, we have made some real progress towards the solution, specifically towards manufacturing more complex, in demand organs. The two most interesting things being organoids (it sounds so sci-fi init?) and organs-on-a-chip (super sci-fi).
Organoids are small 3d organs that are similar to organs in our body, but they aren’t the same. The main differences being size (The volume of 2000 kidney organoids is currently equal to 1 mature kidney) and lack of vascularisation. The two are actually linked. The organoid is limited in the size it can grow (and survive), because it does not have a vascularisation system that can keep it alive.
Brain organoids in a petri dish - Source
As for organs-on-a-chip, they are what they say they are.. to a certain extent. They are 3D cells of an organ on a chip. The Wikipedia definition can be found down below (I have also added a “plain english” translation of it, for those that need it - small plug to jargon - a tool my friend and I built that converts jargon to - you guessed it - plain english )
Organ-on-a-chip - Source
What the converter doesn’t get quite right is its insinuation that medical treatments no longer need to be studied on animals and their tissue, that’s just not how it works (yet). The body is a system that has parts very heavily dependent on each other. So just because you tested a drug on a kidney chip, or even a full bioartificial kidney organ, does not mean it will react the same way in a human's body because 1) for now, we can’t direct treatment to only one organ and 2) because we can’t do that we don’t know how the desired organs interaction will have an effect on it, or other organs its connected to within the body.
That being said, both organoids and organs-on-a-chip are still pretty useful. For starters, they are still both used to test medical treatments as they still provide some useful insight into the treatments potential efficacy and toxicity. They can also be used to generally observe how certain parts of an organ work. So, still useful all round, just not quite there yet.
How do we go about creating them today?
For now, there are 3 major overarching methods; fully automated, semi automated and hand manipulated. Within these 3 categories there are different approaches, which I will be going into just a little bit here, but will expand upon more in upcoming posts.
Fully Automated
Arguably the most popular example of fully automated organ manufacturing is 3D bioprinting. As a brief overview, bioprinting is a form of additive engineering, which is the process of creating things by adding one layer at a time. (As mentioned throughout this post, I will be going into more detail in subsequent posts, so don’t worry about the details for now )
Advantages
Outside of the common advantages that all of these methods share (personalised solutions, less animal testing, reduced transplantion wait times etc), the main advantages of this method are scalability, speed and accuracy.
Scalability. As with anything that’s automated, it becomes easier to replicate.
Speed. The natural development cycle of any organ takes several years. The use of bioprinting dramatically shortens that (for example, the transplanted artificial bladder previously mentioned, was bioprinted and took 2 months to do so). Which contributes massively to a better quality of life for patients (shorter times on medication and external artificial organs).
Accuracy. Naturally, when a machine is left to execute a set of instructions, assuming that the instructions given are correct and tested, it will perform those exact instructions, reducing room for error when executing. This helps reduce quality variation amongst productions, and therefore reduce time spent rectifying or re-doing faulty products - in this case organs.
If it’s made from the patient’s own cells then no immunosuppressants will be needed
Organ supply for patients is not restricted by the number of living or deceased beings willing to donate
Disadvantages
Bioprinting is expensive! Although the general cost of 3D printing is coming down, it’s still not affordable enough that it won’t leave a dent in your pocket, particularly personalized bioprinting.
Although automation helps reduce human error, it unfortunately doesn’t mean it’ll be error free. Sometimes machines act weird (especially hardware). For other products that may not be a big deal, but with organs that are intended to go into a living human, that could have a real impact on them. So it’ll need to be essentially error free, or at least alert operators when something isn’t quite right, no matter how tiny that detail is.
For now, the issue of vascularisation and other similar networks have not been solved, meaning any organ printed now won’t be fully functional (it is not entirely clear to me how the bioprinted bladders got around this problem, but once I find that out I’ll let you guys know).
Semi Automated
An example of a semi automated process would be rotational combined molding. The official Wikipedia definition is as follows:
“Rotational molding (BrE: moulding) involves a heated hollow mold which is filled with a charge or shot weight of material. It is then slowly rotated (usually around two perpendicular axes), causing the softened material to disperse and stick to the walls of the mold.”
Or, in plain english:
Diagram of rotational combined molding - Source & more images of this process here
Advantages
In comparison to the natural formation of organs within humans (which takes years), it has a much shorter development time, which in turn reduces wait time for transplant patients
Assuming it is made of the patient’s own cells, then the need for immunosuppressants will be eliminated
Organ supply for patients is not restricted by the number of living or deceased beings willing to donate
Disadvantages
Given its dependency on extremely accurate heating and cooling, this leaves a lot of room for error, particularly material degradation or shape deformation.
It requires having the entire scaffold of the organ done, including intricate systems such as the vascular system, which we already acknowledged as quite difficult.
As this is a semi automated process, room for human error is naturally high.
It also comes with relatively high operational costs given the human labor involved.
Hand Manipulated
The two most well known examples of this are decellularized organ regeneration and external growing.
Decellularized organ regeneration is the process of removing cells from an already grown organ (either in a human or animal), and using the organ's shape as a scaffold. The patient's cells are then injected. Because these scaffolds do not have the original host cells, they are not subject to rejection from the patient. The issue of vascularisation is also not an issue in this case, as the organ will already be vascularised as it would have gone through embryogenesis.
Pig heart decellularized (stripped of its genetic material) - Source
Advantages
The biggest advantage of this method is that both the internal and external structure are already made perfectly.
From a transplantation point of view, it could also bring the advantage of shorter wait times. For example, in the case of kidney patients, if organs were harvested from pigs then at best they would wait around 12 - 18 months, which is much better than the long waiting times.
As the organ will be stripped of its original DNA and replaced with the patient in question’s cells, this eliminates the need for immunosuppression, drastically improving the quality of life for patients.
Disadvantages
In a world trying to decrease its dependence on animals for its own consumption, growing animals for the sake of harvesting their organs could bring up some ethical issues.
Where the desired organ can not be grown and harvested from another animal in a short amount of time, this maintains the same supply issues we have today.
If the organ comes from another animal, there is a chance that an infection / virus specific to that animal may be transferred to the host, as was the case with a patient who had a pig heart transplant but unfortunately died from a pig virus. In an extreme case, it could lead to the spread of a virus amongst humans that have deadly effects - HIV and COVID-19 are an example of this
There may be differences in the structure of the donor organ, when comparing it to the host's original organs. When studying disease development, especially when taking a personalized approach, this may cause some discrepancies.
Growing organs externally is the process of performing organogenesis outside of the body.
Advantages
Because it goes through organogenesis, the issue of internal structures such as the vascular system, will not be an issue.
Ideally, this would be grown from the patient's own cells, which will mean the patient will not require immunosuppressant drugs.
Disadvantages
To make a fully developed organ takes a long time, much longer than 12 months. Although this is shorter than current wait times for transplant patients, it is still longer than other solutions mentioned.
It is extremely difficult to replicate organogenesis outside of its natural habit (a fetus within a womb)
What should I expect from this newsletter?
I plan on sharing my technical notes on different scientific and engineering subjects related to this topic, the latest developments within the space and simplified technical papers. I will also be continuing my kidney series here.
I should make clear that these are all ideas I am experimenting with, so they may get scrapped! If you have anything else you would like me to cover in this newsletter please email me adaobiadibe23@gmail. Or if you just want to chat about ideas discussed here that’s fine too :)